|
This week, we've been talking about numbers (ah!) and learning about how to express significant figures. If you need more explanation about them and how to express them in calculations, refer to pages 66-70. We also talked about the importance of scientific notation because in chemistry, we talk about really, really big things and really, really small things (check it out!). Any questions about scientific notation, check out pg. 65.
0 Comments
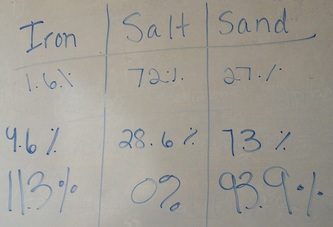
Below is the lab data for the lab where we separated iron filings, salt, and sand from a heterogeneous mixture. Each group of data is from a different period so make sure you get the right one! You'll need to compare your data with the classes in terms of precision and compare your data with the actual data that I will give you here: Iron: 9.2% Salt: 15.6% Sand: 75.2% If you missed the lab, the YouTube video below gives a quick overview of what was done in class. The only difference is that we did not evaporate the water but rather let the sand dry to get the mass of the sand to calculate the mass of the salt. If you need data, please ask a lab partner for data. Thanks! After reviewing heterogeneous and homogeneous mixtures and compounds and elements, we talked about physical and chemical properties as a way to describe substances. If you need to find those definitions, they are on pg. 40 and pg. 53 of your textbook. Then, we learned about the changes that substances can undergo, whether they are physical changes or chemical changes (on pg. 42 and 53 of your textbook). Despite the change that happens to a substance, mass is always conserved though! That's because of the law of conservation of mass (pg. 55). To see how molecules react in the states of matter, which are physical changes, you can play around on this simulation! To solidify (ha!) our understanding, there was a worksheet we did too!
Today, we were introduced to what chemistry is and how scientists classify matter. Check out the Powerpoint presentation to stay caught up!
On Friday, we started to investigate how we know the things we know. We did this by designing a lab to test how we can make bubbles with different qualities using different ingredients in our solutions such as salt or sugar in addition to dish detergent.
Today, everyone was able to get their hands on the supplies and actually play with some bubbles! To check out what the homework for this lab is, click on the assignments tab! |
Archives
May 2015
What Can I find here?This blog will include regular updates of what goes on in class. Check it often to make sure you're on track to pass with flying colors! Categories |
||||||||||||



 RSS Feed
RSS Feed
