| 9.30.13.ppt |
|
We went over the periodic trends of ionization energy and electronegativity. We also dove into learning about how ions combine to make ionic compounds. Check the slideshow for all the information you'll need!
0 Comments
We clarified our understanding of ions through Lewis dot structures. Make sure you understand it by checking out the slideshow below!
We are jumping into learning about ions and what ions form from certain elements according to their valence electrons. No powerpoint for today because it was a short day and we were testing clickers but check back tomorrow!
Today, we started a new section learning about bonding of atoms. We had a review of what we have learned about atoms in terms of subatomic particles and the electrons in different atoms. Then we built on that knowledge to just talk about valence electrons and how to show the electrons that are on the outermost level of an atom. Check out the powerpoint presentation for more information. The pages that the information is on can be found in the presentation. We also have a test on Friday! The test study guide is below.
Today, we finished up learning about electron configuration and were able to review it. An atom project was introduced! If you need the instructions for it, check below. The quiz 4 review is also below. AND! The powerpoint for today is there as well. Enjoy! If you need tutoring, tomorrow during 5th period is Ms. Au-Yeung's tutoring session! Meet in room 402. If you need to make up the Flame Test lab, make sure to come to 402 during 5th period tomorrow!
We debriefed the lab today and talked about ions. For the definition of an ion, refer to page 172 in your textbook. We are also continuing to explore electron configuration (pg. 133) and learned about atomic spectra as a result of gaining energy. Check out the powerpoint for today!
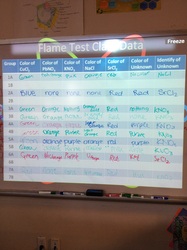
Today, we did a flame test lab to investigate how to identify an unknown solution. This is based off of an understanding of different energy levels of atoms of different elements. The class data for each period is posted below - Make sure you're looking at the right period! If you were in groups 1, 3, 4, or 5, you will need to only look at that data. If you were in groups 2, 6, or 7, you will only compare your data to those groups. If you missed the lab, don't worry! You will have an opportunity to actually do the lab yourself. The powerpoint for today is posted below.
We are building on our understanding of the atom by talking about electrons and where they can be found. First, we need to know how to identify the region (or the address) of the electrons of particular atoms. So! To do that, we need to learn about electron configuration. Please look at Figure 5.7 on pg. 133 to get an idea of the levels in which electrons start to build around an atom.
We're learning more about how the periodic table is organized! If you need a copy of the periodic table, there is a copy of it below. Please outline the periodic table with where the metals, nonmetals, and metalloids are found. A periodic table showing that is found on pg. 158. We also learned about the properties of each of these larger groups which can be found on pgs. 158-159. On the periodic table below, we will be following the topmost numbers of each group (column of the periodic table). To better familiarize ourselves with the names of the groups, here they are below: Group 1 - Alkali metals Group 2 - Alkaline earth metals Groups 3-12 - Transition metals Group 17 - Halogens Group 18 - Noble Gases. Check out the Quiz 3 Review below too!
If you need extra help understanding the parts of an atom, check out this simulation!
|
Archives
May 2015
What Can I find here?This blog will include regular updates of what goes on in class. Check it often to make sure you're on track to pass with flying colors! Categories |
||||||||||||||||||||||||||||||||||||||||||||||||||||||


 RSS Feed
RSS Feed
